|
With skin, hair, and clothing particles coupled with constant movement, humans contribute roughly 75-80% of contamination in a cleanroom. It might seem trivial to be so concerned with where people are, but personnel is a top contributor to cleanroom contamination. GMP Annex 1 specifies that, for a cleanroom to be at-rest, the personnel must be gone for at least 15 minutes.įinally, when a cleanroom is operational, it means that the cleanroom is set up, in operation, with the appropriate number of personnel present. When a cleanroom is at-rest, the cleanroom needs to be completely assembled and operational, but no personnel can be present. But there is no equipment, machinery, materials, or personnel. This includes all services being hooked up to the cleanroom. When a cleanroom is as-built, that means all the infrastructure is in place. There are three different types of cleanroom occupancy states: as-built, at-rest, and operational. On the other hand, GMP Annex 1 has 4 grades (Grade A, B, C, and D, where Zone A is the cleanest). ISO 14644-1:2015 has 8 classifications of cleanrooms (ISO 1 through 8, where ISO 1 is the cleanest and ISO 8 is normal room air). There are two primary classifications we use: ISO 14644-1:2015 and GMP Annex 1 2020 draft. Even if you have a system of remote particle counters in place to monitor your cleanroom(s), you will need to test more areas during the classification process. Typically, you should use a portable particle counter when classifying your cleanroom. These locations should be chosen through a strategic risk based approach that we will cover later in this article. You can attain your classification through measuring the particle levels in different areas of your cleanroom. Often, these regulations are put into place by the Food And Drug Administration (FDA).Ī classification lets people know your cleanroom is a certain level of clean. Cleanroom Classification BasicsĬlassifying cleanrooms is essential for the peace of mind of your distributors and, quite likely, required. This article covers cleanroom classification bases, definition of cleanroom occupancy states, your risk based approach to cleanroom classification, and reclassification periods as defined by popular certifications. They pertain to cleanroom classifications, of which there are several requirements. They were developed by pharmaceutical industry professions for pharmaceutical industry professionals! These standards aim to improve compliance, quality, efficiency, and cost reduction. Following GAMP best practices, manufacturers are able to track and record their processes for certification. GAMP certifies a system that produces quality products following a life cycle model. GAMP (Good Automated Manufacturing Practices)is a system designed specifically for suppliers and users in pharmaceuticals. 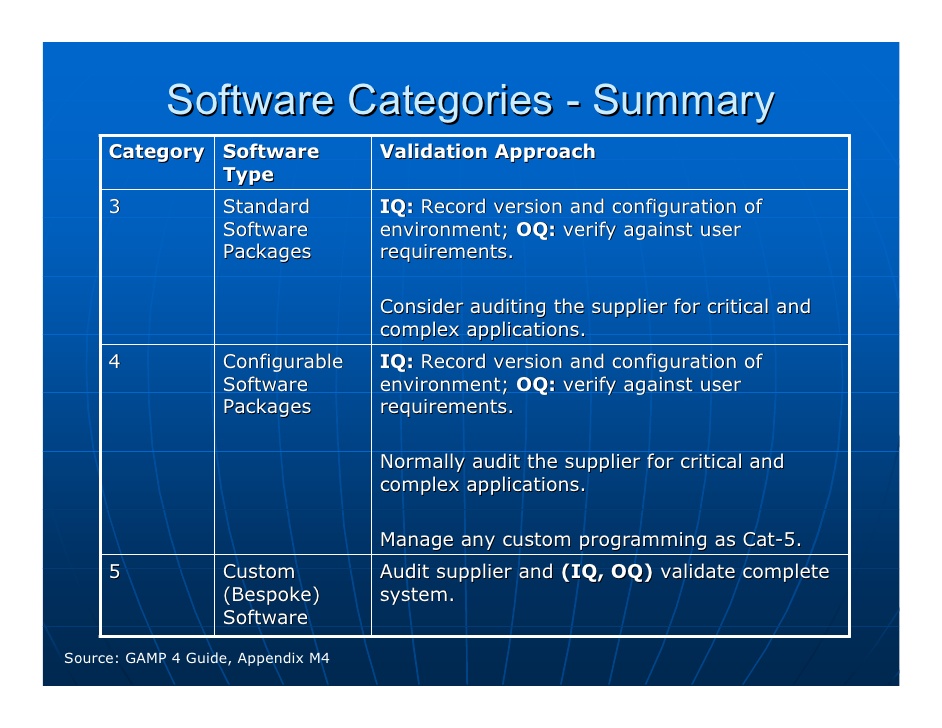  
GAMP was designed to address these needs. This is especially true in the area of computerized system compliance and validation. In an ever changing world, we are constantly seeing new regulations and expectations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
